ISO/IEC 27001 made practical
With BPA, transform ISO requirements into efficient and auditable processes
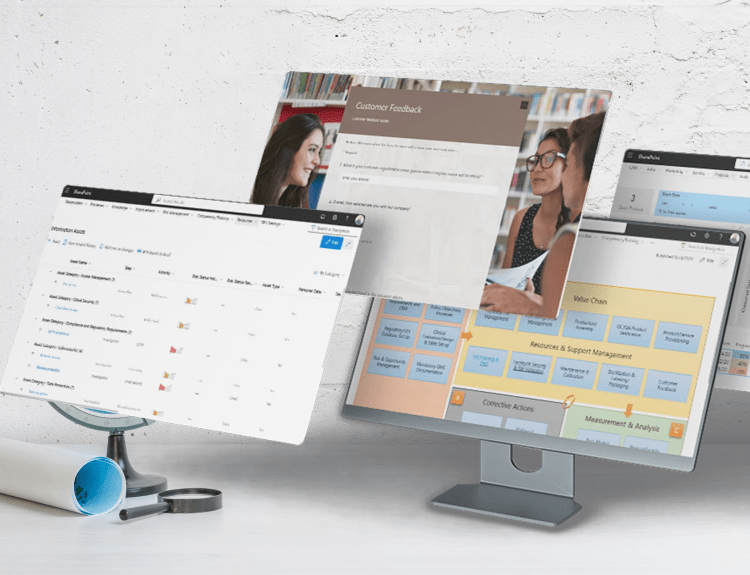
Implementing and maintaining an Information Security Management System compliant with ISO/IEC 27001:2022 is a strategic priority for organizations seeking to protect sensitive information, manage cybersecurity risks, and meet growing regulatory and customer expectations.
BPA Solutions helps transform ISO 27001 requirements into practical, efficient, and auditable processes. Through its AI-driven BPA eQMS, organizations can manage the full ISMS lifecycle with confidence, from governance and risk assessment to document control, audits, training, and continuous improvement.

Download our guidebook to gain clear guidance on ISO 27001 fundamentals and understand how BPA supports each clause with a detailed feature-to-standard mapping.
Discover how ISO 27001 compliance can be embedded into daily operations, supporting certification, long-term compliance, and a stronger information security posture.
KOMMENDES WEBINAR
Driving trust and growth with ISO 27001: from audit readiness to operational excellence
March 17 – 16.00 CET
With BPA unleash the full power of Microsoft 365
to streamline your quality processes without disrupting user habits
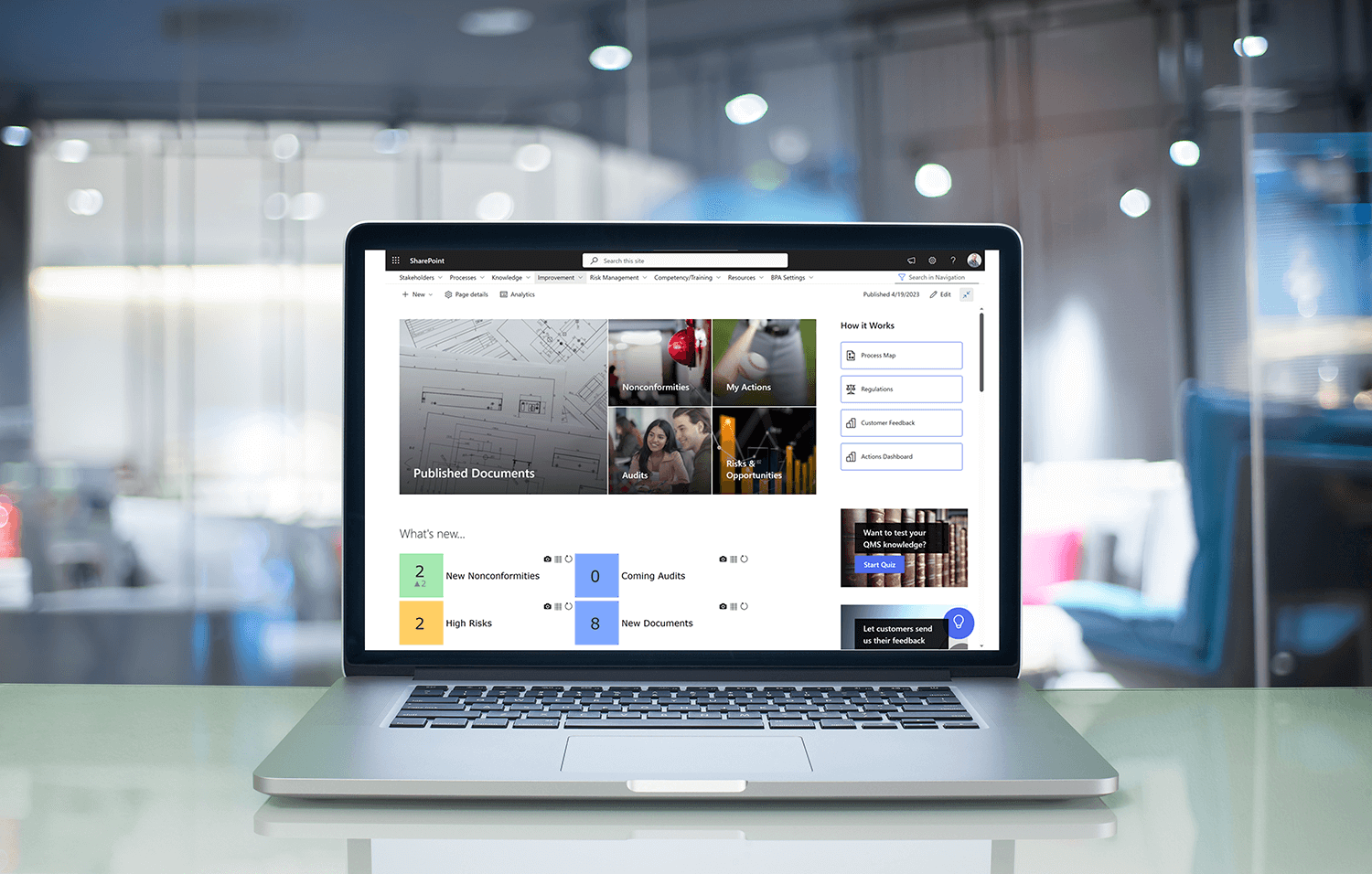
What are BPAQuality365 and BPAMedical365?
- BPAQuality365 and BPAMedical365 are two powerful, Microsoft 365-based eQMS (electronic Quality Management System) platforms, designed to suit different regulatory and operational needs, but both with a strong focus on usability, Automatisierung, Und compliance. Intuitive and agile, they run directly on Microsoft 365 (SharePoint, Teams, Power BI, etc.) and support secure deployment, including in highly secure environments (such as GCC High) to meet strict data-control requirements. Learn more about why you should choose Microsoft 365 for your QMS >
- Covering everything from document control, audits, non-conformances, risk and training management, they are designed to help companies comply with common quality and risk standards (like ISO, FDA, DSGVO, EU MDR) while helping users navigate quality tasks more efficiently.
- BPAMedical365 is specifically built for regulated life sciences, medical devices, pharma and healthcare industries. The platform is fully compliant with GxP guidelines, ISO 13485, EU MDR, FDA 21 CFR Part 11, offering features like electronic signatures, access control, and complete audit trails. Coming with a Validation Toolkit (SOPs, test scripts, validation templates) to accelerate regulatory validation and reduce validation cost, BPAMedical365 is a pre-validated eQMS solution. Learn more about the Validation Toolkit >
How long does it take to implement BPA?
- Most clients go live in 3–4 months with less than 10 days of BPA expert support. Our streamlined delivery ensures quick time to value and minimal disruption for your team.
- Clients typically allocate around 10% of their project team’s time over the 3–4-month implementation. BPA manages the setup, training, and onboarding to minimize internal workload.
- We follow a standardized quick-start approach including weekly sessions, knowledge transfer, and out-of-the-box configuration. Prebuilt workflows accelerate ROI, and your team becomes autonomous rapidly.
Does BPA integrate with Microsoft Dynamics 365 and other ERP/CRM systems?
-
Absolutely. BPA integrates natively with Microsoft Dynamics 365, including Business Central Und Finance & Supply Chain Management (F&SCM), through our dedicated QMS connector. This enables synchronized records, automated workflows, unified master data, and real-time visibility across quality, operations, and ERP processes, creating a fully connected and efficient Microsoft-based ecosystem.
-
BPA also integrates with third-party systems like SAP Und major CRMs/ERPs. Because BPA runs on SharePoint and Microsoft 365, external integrations work like any standard SharePoint connection, supported by numerous connectors and customization options. Whether you need synced data, automated quality workflows, or deeper supply-chain connectivity, BPA helps you build a seamless, high-performance QMS environment tailored to your business.
Is BPA suitable for multi-site or global deployments?
-
Absolutely. With Microsoft 365 as the foundation, BPA supports multi-site operations, distributed teams, and global compliance needs with consistent processes and centralized control.
-
BPA is designed for high scalability, allowing you to start with the modules you need today and easily expand as your organization grows. Additional out-of-the-box modules, user licenses, Power Extensions, and BI reporting can be activated quickly and seamlessly, ensuring your eQMS evolves at the same pace as your quality and compliance requirements.
What modules and Power Extensions are available with BPA?
-
BPA includes a broad suite of prebuilt quality modules (documents, risks, audits, training, nonconformities, CAPA, equipment, suppliers, and more) covering the full quality lifecycle. To enhance usability, BPA offers Leistungserweiterungen, including mobile apps ideal for field workers, automated workflows, advanced reporting with Power BI, and rich user experiences designed to simplify and accelerate compliance activities.
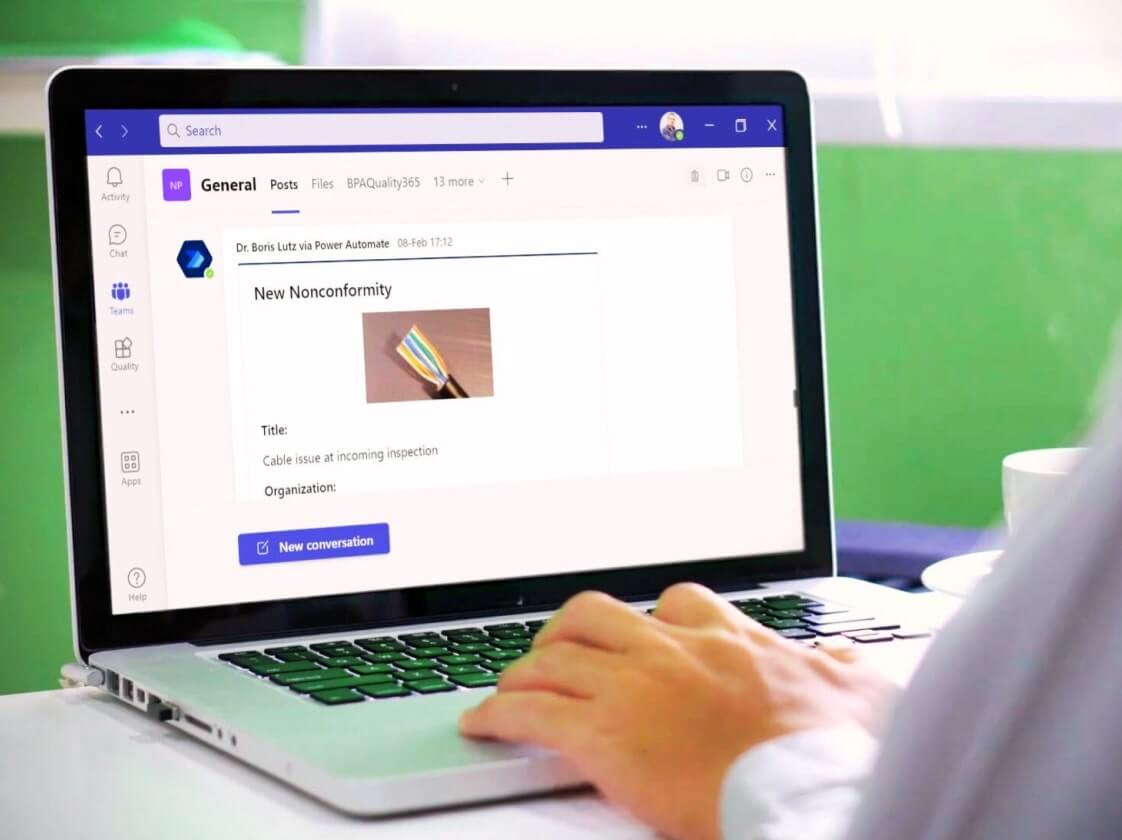
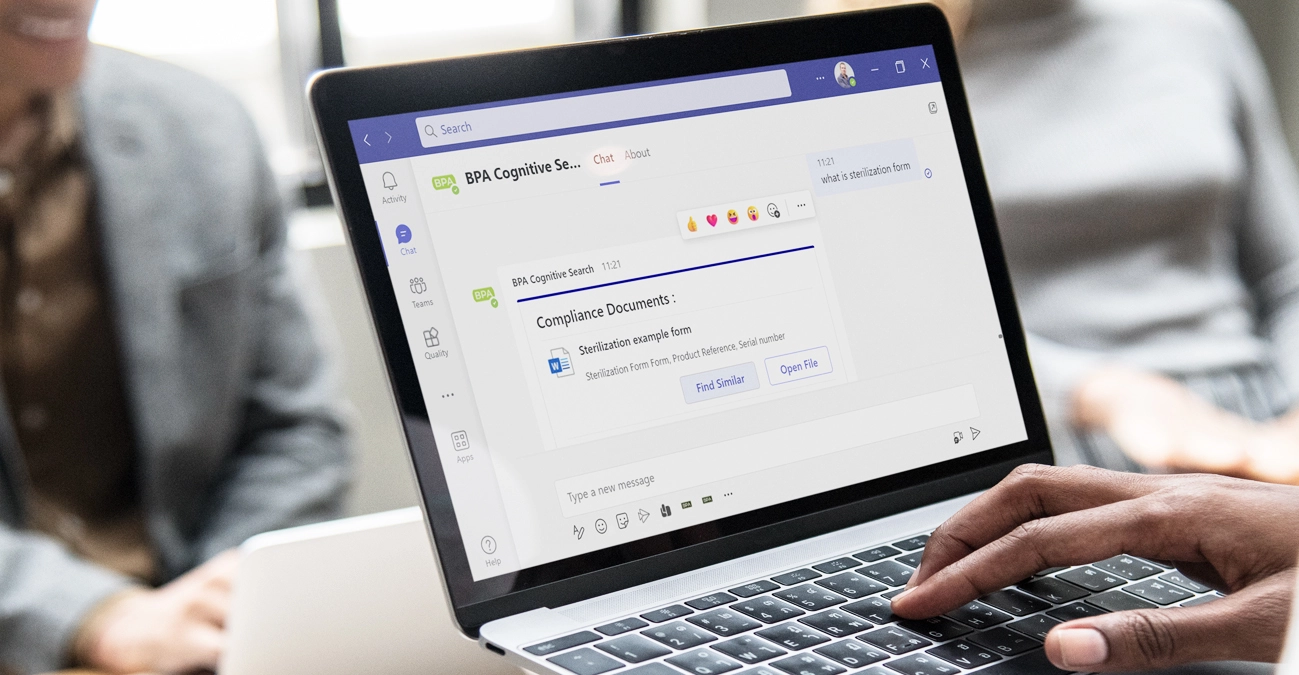
How is AI integrated into BPA and how does it leverage data to ensure security and compliance?
-
BPA’s QMS comes with powerful, safe, and productivity-boosting GenAI built for quality and compliance. BPAQuality365 and BPAMedical365 include built-in GenAI capabilities through Q-Pilot, BPA’s secure, enterprise-grade virtual agent. Q-Pilot allows users to instantly find the right procedures, records, risks, or training information simply by asking natural questions, dramatically reducing time spent searching or navigating the QMS. It also assists quality managers with everyday tasks such as summarizing content, guiding workflows, and improving overall productivity. Learn how Q-Pilot can assist you >
-
Security and compliance are foundational to BPA’s AI integration. All AI processing happens within your Microsoft 365 environment, and no data is shared externally or stored outside your control. This secure-by-design approach ensures that AI benefits are delivered without compromising data privacy, regulatory requirements, or access governance, giving Quality, Compliance, and IT leaders full confidence in both innovation and security. Learn more about BPA’s secure-by-design approach >
Wo werden die Daten gespeichert, und wer kontrolliert sie?
Your data is securely stored in your own Microsoft 365 environment, directly on SharePoint and fully controlled, governed, and monitored by you. Because BPAQuality365 and BPAMedical365 run natively within your Microsoft tenant, no data is transferred, hosted, or duplicated on external systems. BPA Solutions never accesses or stores your information.
This “your environment, your data” approach ensures security, compliance, and data-sovereignty. Combined with BPA’s secure-by-design architecture, your quality data stays protected, private, and fully compliant while benefiting from the power of Microsoft 365. Learn more about BPA’s “your environment – your data” approach >
Do you support high-security environments (e.g. GCC High)?
Yes. BPAQuality365 and BPAMedical365 fully support high-security Microsoft cloud environments, including GCC High. Because our eQMS is installed directly inside your own secure Microsoft 365 tenant, you benefit from your existing government-grade security, compliance, and data-residency controls, without relying on external hosting.
Our GCC High–ready architecture ensures that regulated organizations, government contractors, and defense-related industries can run a modern, Microsoft-based eQMS while meeting strict security and regulatory requirements. Learn why BPA is a GCC High Cloud-ready eQMS solution >
What security measures are in place?
BPAQuality365 and BPAMedical365 rely on the enterprise-grade security of your Microsoft 365 environment, ensuring your eQMS meets the highest standards for data protection and regulatory compliance. All security measures are built-in, secure-by-design, and governed entirely by your tenant. Key protections include:
-
Data encryption at rest and in transit using Microsoft 365’s advanced security framework.
-
Multi-Factor Authentication (MFA) and conditional access policies to prevent unauthorized access.
-
Granular permission and role-based access control, ensuring users only see what they are authorized to see.
-
Complete audit trails, secure electronic signatures, and validation-ready controls for regulated industries.
-
Secure-by-design AI features, ensuring no data leaves your Microsoft tenant and all AI interactions remain private and compliant.
This architecture delivers a highly secure, compliant, and region-aligned eQMS, ideal for organizations with strict quality, regulatory, or data-sovereignty requirements.
Weitere Demo-Videos ansehen
Füllen Sie das Formular aus und erhalten Sie exklusiven Zugang zu einer Vielzahl von Video-Ressourcen über unsere Lösungen, Module, Erweiterungen, Neuigkeiten und Animationen.
Über 200 begeisterte Kunden vertrauen uns
die mit BPA Solutions weiter wachsen
Sehen Sie aus erster Hand, was BPA Solutions zu bieten hat
"Wir waren in der Lage, unsere 1200 Qualitätsdokumente erfolgreich in ein elektronisches, vollständig verwaltetes System mit Versionskontrolle und elektronischen Signaturen zu integrieren - etwas, das wir für unsere regulatorischen Anforderungen implementieren müssen.....".
©2026 BPA Solutions SA