Announcing Software Validation Package with BPA Apps

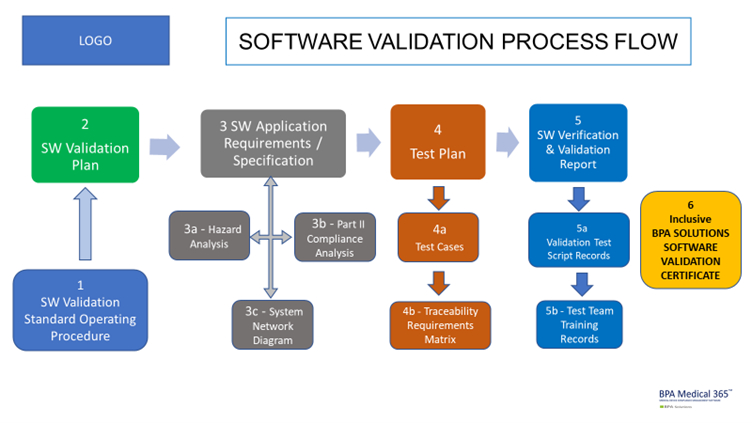
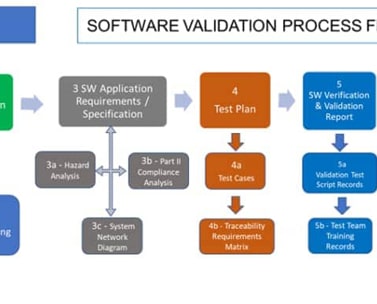
The BPA software validation package includes documents mentioned in this process flow.
In regulated industries, it’s required to validate any software which has an impact on the delivered products. The concerned software are typically used for design and development, production, monitoring and measurement, quality and document management activities.
According to ISO 13485:2016, MDF and FDA, evidence is required that the software specifications are continuously in accordance with user requirements.
For many reasons, software validation is beneficial for organizations. It saves money by discovering weaknesses or defects before installing the software (hazard analysis). It helps to select the most suitable software and complete the project on time, without exceeding the budget.
Software validation requires several questions to be answered, like:
- what is the purpose of the software?
- what makes you think that the software is functional?
- can you tell when the software is not working?
- what are the risks of software shutdown and what will be the impacts?
As a Quality, Risk and Document Management editor, our software is concerned by validation. We are currently working on a software validation package to help clients with the BPA software validation. The package includes several documents and templates mentioned in the above process flow.
The BPA software validation package is the most economic option for organizations to validate our software. Still, complete test cases and evidence need to be prepared by the client, based on the deployed software modules and configurations done.




