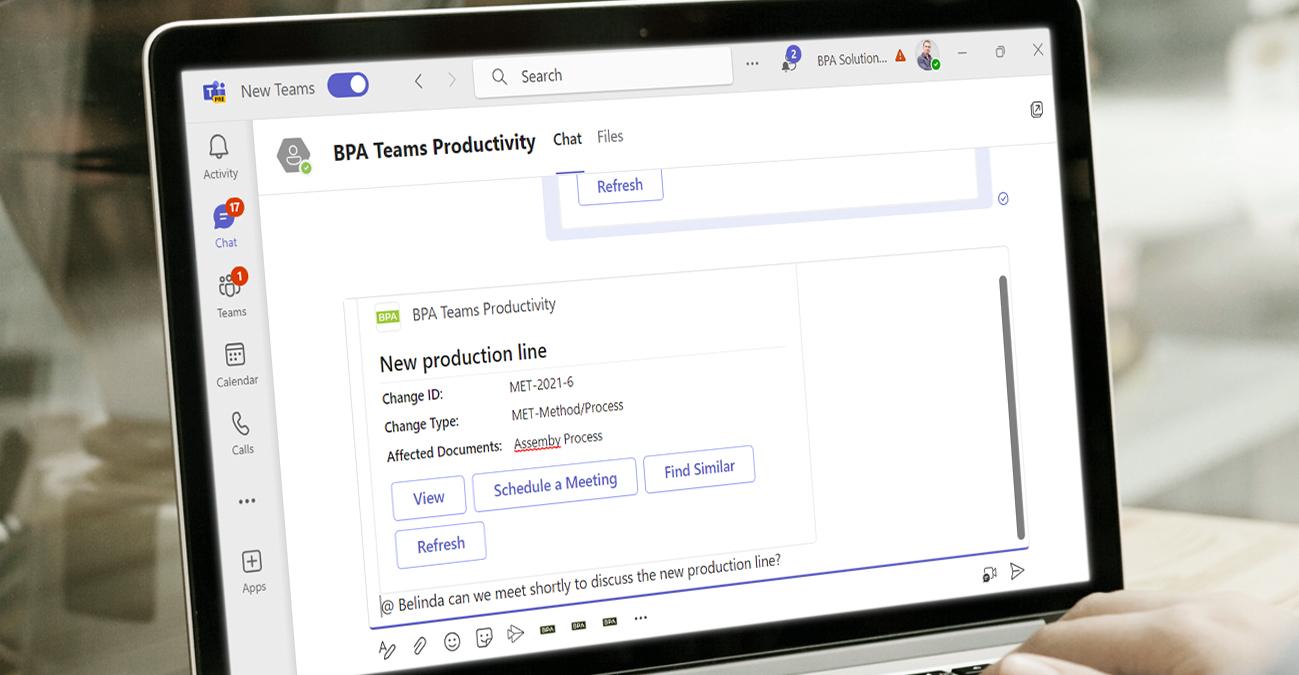
Secure eQMS adapted for Pharma, Biotech and Medtech markets
BPA eQMS software solutions assist organizations in the pharmaceutical, Medtech and Biotech sectors by supporting quality management, document control, compliance management, risk management, training management, and change control processes. The corresponding solution modules BPA offers fully leverage Microsoft 365 technologies and enable such industries to significantly improve operational efficiency, ensure regulatory compliance, mitigate risks, and enhance patient safety and product quality.

Access additional resources for this solution
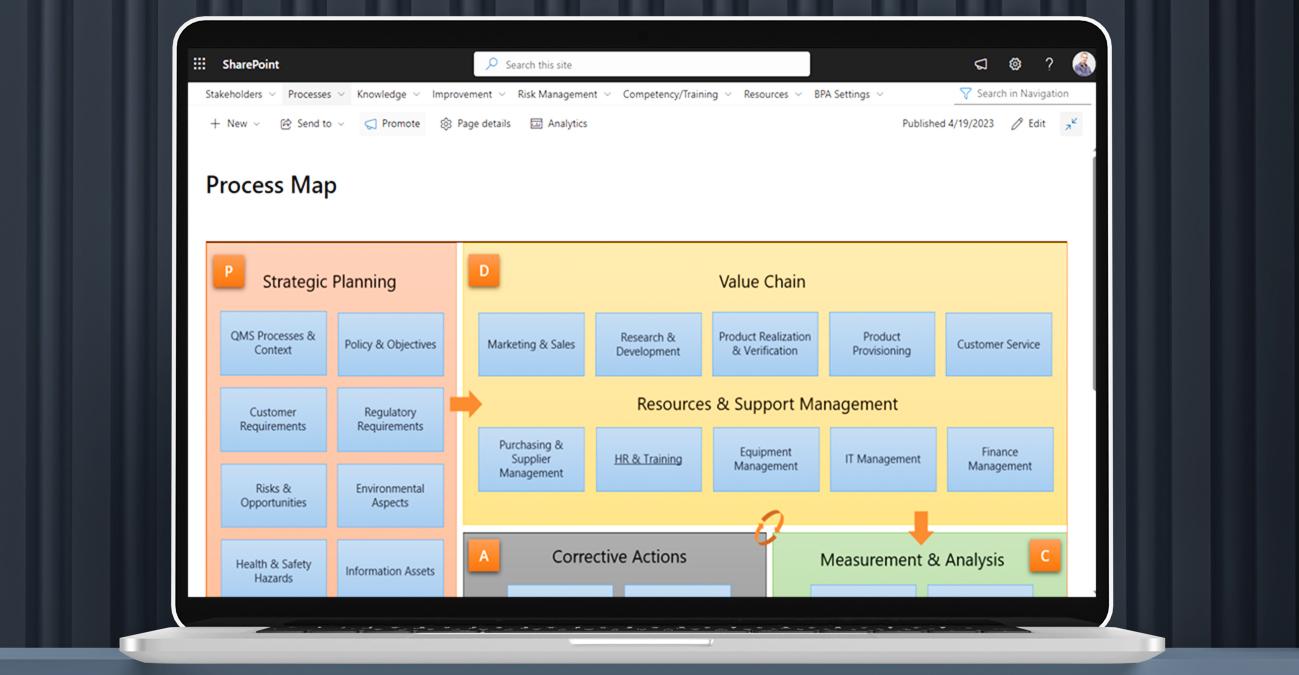
Effective quality management
BPA’s eQMS helps Pharmaceutical, Medtech and Biotech companies effectively manage their quality management processes. It allows them to enforce quality standards, monitor deviations and non-conformances, and implement corrective actions. The software ensures compliance with all regulatory requirements, reducing the risk of product recalls or regulatory penalties, and improving overall product quality.
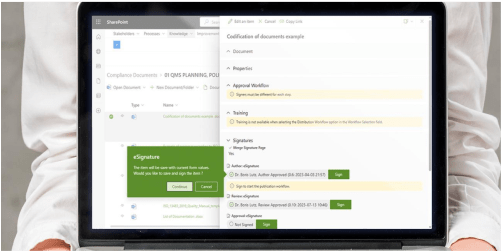
Efficient document control
BPA’s QMS software facilitates efficient document control by providing a centralized repository for storing and managing critical documents such as standard operating procedures (SOPs), protocols, and technical files. The software enables version control, document approval workflows including e-signatures, and access permissions, ensuring that employees have access to the most up-to-date and accurate information.
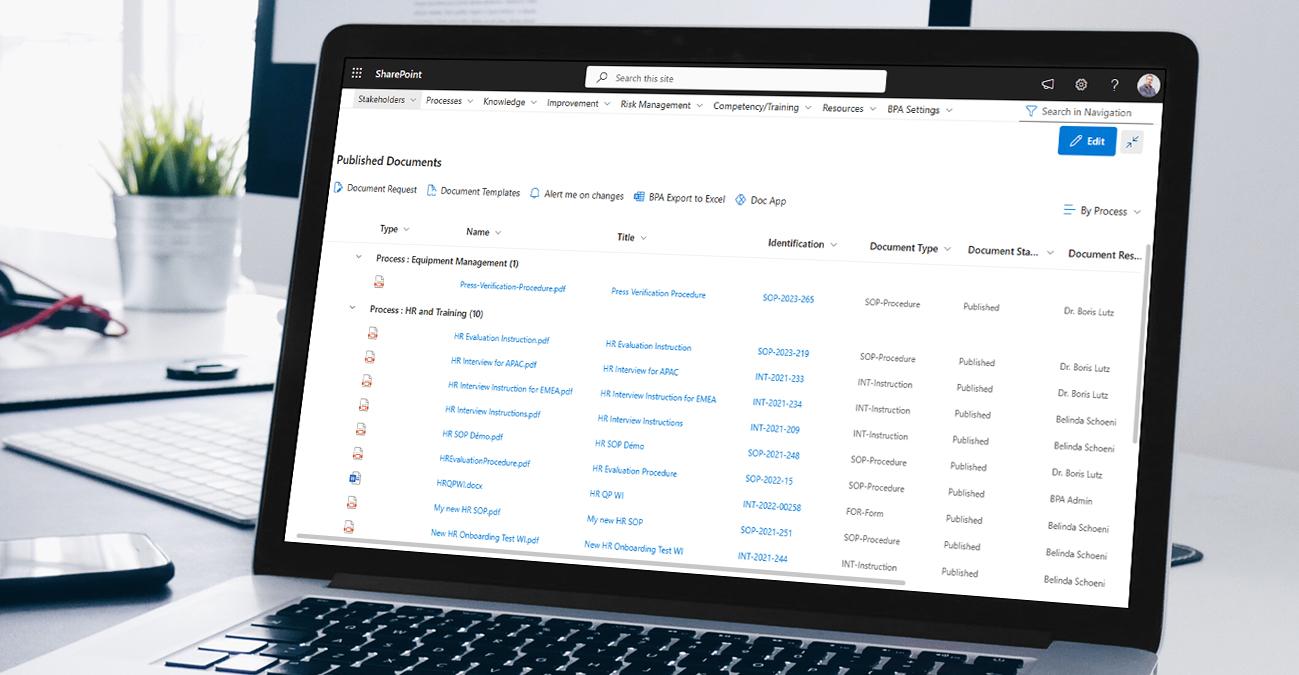
Sustainable compliance management
BPA software supports companies in the Pharmaceutical, Medtech and Biotech sectors adhering to strict regulatory requirements. It helps manage document and compliance processes, including regulatory audits and inspections. The software assists in maintaining compliance with Good Manufacturing Practices (GMP) and Good Laboratory Practices (GLP). This ensures conformity, reduces risk, and strengthens reputation.
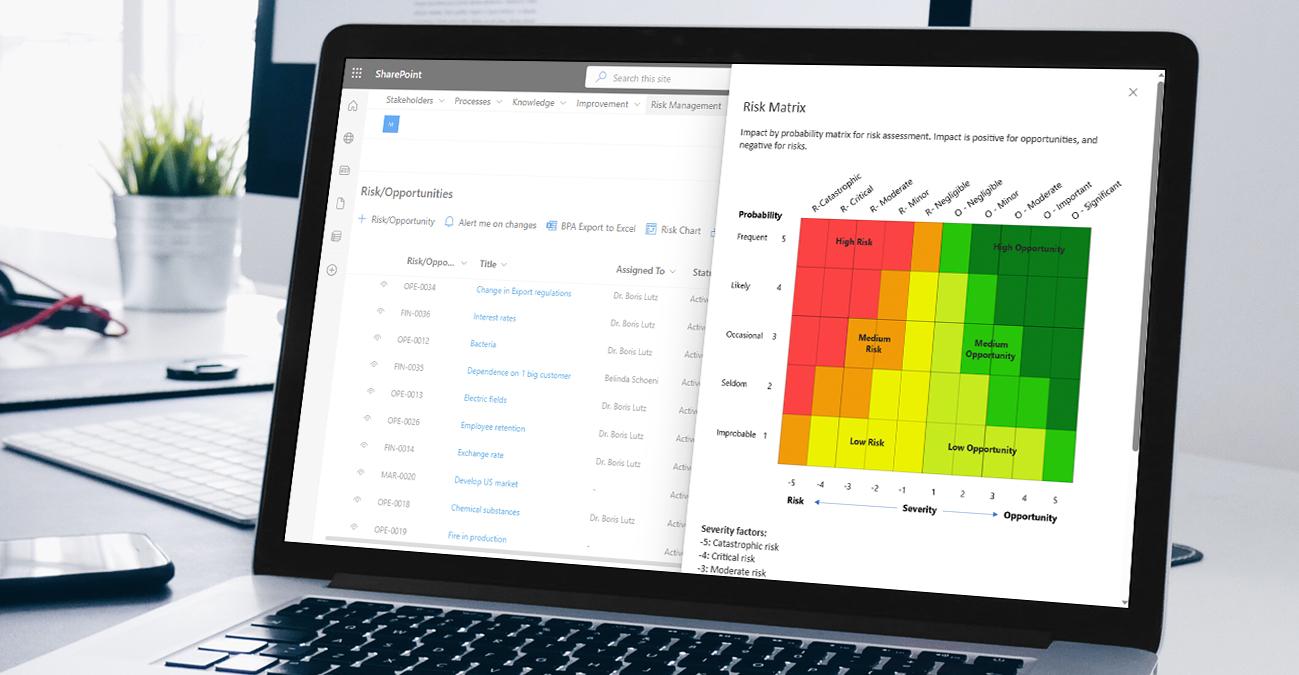
Reliable risk management
BPA software solutions enable organizations to reliably identify, assess, and mitigate risks associated with their products and processes. It provides tools for adequately documenting and analyzing risks, implementing risk control measures, and monitoring risk mitigation actions. The software allows companies to proactively manage risks, minimize potential harm to patients, and maintain product safety and efficacy.
Thorough training and competency management
BPA software strengthens training and competency management. It allows organizations to create and deliver training programs, track employee training records and certifications, and evaluate competencies. The software ensures that employees are adequately trained and competent to perform their tasks, reducing errors and ensuring compliance with training requirements.
Structured change control
BPA’s QMS platform facilitates effective change control across sites and departments. It provides a structured and documented approach to managing changes in processes, systems, or products. The software helps evaluate change requests, assess potential impacts, implement changes, and track their effectiveness. Properly manage, validate and document changes, minimizing disruptions and maintaining product quality and safety.
Microsoft 365
The best technology for your QMS
Secure
Microsoft 365 provides the highest security for your QMS data. You have full ownership and control of your data
Trusted
Profitable
Lower costs leveraging acquired Microsoft 365 tools for your QMS, as SharePoint, Teams, PowerBI, etc.
Trusted by 200+ delighted customers
that keep growing with BPA Solutions
Uppsala Monitoring Center
Uppsala Monitoring Center and BPA Solutions | #1 eQMS powered by Microsoft 365
See firsthand what BPA Solutions has to offer
“We were able to successfully integrate our 1200 quality documents into an electronic, fully managed system with versioning control and electronic signatures, which is something that we have to implement for our regulatory requirements.....”