Symbios Success Story
Who is Symbios?
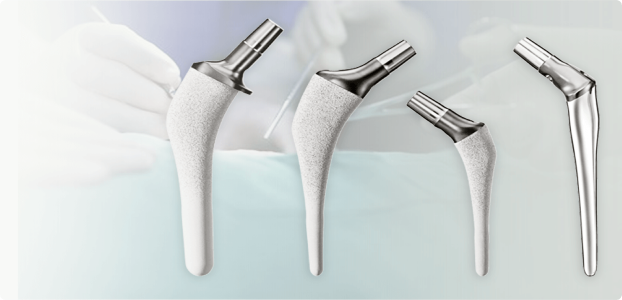
Symbios is a Swiss medical technology company, and the leader in individualized orthopedic implants for hip and knee joint replacement. With the goal of designing and manufacturing individualized implants that adapt perfectly to each patient’s unique anatomy, Symbios provides products that are better adapted to patients while allowing for a significant reduction in stocks and instruments for healthcare facilities.
Who are you and what do you do?
My name is Sooah Barbieri I work for Symbios Orthopédie SA in Yverdon, Switzerland.
I am a Quality System Specialist and Good Manufacturing Practices trainer. Symbios Orthopédie produces individualized hip and knee joint replacement implants, we make all our products in Switzerland. We have approximately 200 employees worldwide, most of whom are based in Switzerland.
Which QMS solution did you implement?
Symbios Orthopédie decided to implement BPAMedical365®, as we were looking for a fully electronic Document Management System for our quality documents. In the future, we will be implementing and testing out the other modules offered by BPAMedical365®, which are risk management, equipment manufacturing management, non-conformities and CAPAs.
Which results were you able to achieve with the BPAMedical365®software?
We were able to successfully integrate our 1200 quality documents into an electronic, fully managed system with versioning control and electronic signatures, which is something that we have to implement for our regulatory requirements.
You were successfully audited by the EU Notified Body. Which key elements brought BPAMedical365® to comply with their regulations?
Since our pilot and roll out in September 2022, we have been audited by four different notified bodies, we have successfully been audited by them with no major/minor non-conformities. For our electronic quality system, we are able to show our versioning control and the capacity to effectively roll out our quality documents with e-signatures.

Medical Devices

