Logiciel QMS médical intuitif
Facilitate compliance with ISO 13485, MDR and FDA with a pre-validated, secure and cost-effective eQMS powered by SharePoint and Microsoft 365 technologies

Intuitive
- Intuitive, easy adoption and quick deployment.
- Environnement familier, pas de perturbation des habitudes.
- Pour tous, sur tous les appareils.
Agile
- Évolue avec votre entreprise.
- Hautement configurable.
- Interactions transparentes.
Rentabilité
- Fully leverages SharePoint, Teams and much more.
- Ready-to-use, pre-validated and secure.
- Les utilisateurs internes/externes peuvent alimenter le SMQ gratuitement.
Offrir une qualité intelligente à tous les utilisateurs, quel que soit l'appareil utilisé
Atteindre l'excellence grâce à un système de gestion de la qualité de bout en bout
- Un abonnement qui intègre plus de 30 modules pour l'amélioration continue et la gestion du cycle de vie du produit.
- An intuitive and robust platform that facilitates compliance with any GxP, ISO, FDA, MDR, 21 CFR Part 11 requirement and more!
Faciliter la conformité grâce à l'automatisation et à la GenAI
- Faciliter la mise en conformité avec des schémas de processus visuels, des règlements et des processus clairement expliqués, et des signatures électroniques solides.
- Réduire le travail manuel et garantir l'exactitude avec des fonctions pré-intégrées d'automatisation des processus et d'IA générative.
Accelerate time to validation and stay compliant
- Deploy faster, stay compliant and seamlessly respond to audits with our Validation Toolkit providing a complete set of pre-defined, regulator-ready validation deliverables.
- Reduce the total cost of validation with a complete and audit-ready documentation suite curated by our expert validation partners.
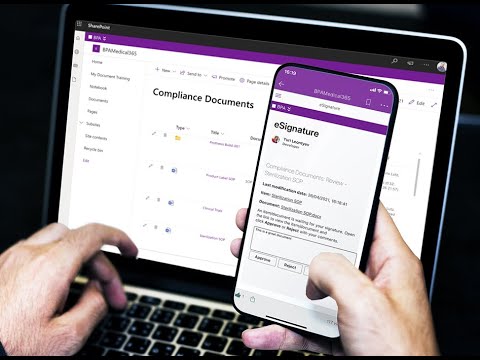
Garantir des approbations valides grâce à des signatures électroniques solides
- Make sure only authorized people can sign documents/forms by using the trusted Microsoft multi-factor authentication system.
- Un moyen fiable d'approuver des procédures ou des dossiers techniques importants.
Satisfaire aux directives GxP et 21 CFR Part 11
- Document retention, access control et audit trail are included in BPAMedical365® pour s'aligner sur les exigences GxP et 21 CFR Part 11.
- Le module intégré de signature électronique offre une sécurité supplémentaire lors de l'approbation des documents.
Impliquer tous les utilisateurs dans le système de gestion de la qualité
- Faire de tous les utilisateurs des contributeurs actifs au SMQ avec leurs outils et dispositifs préférés (cols bleus et blancs, personnes sur le terrain, etc.).
- Ne modifiez pas les habitudes des utilisateurs et assurer l'adoption with SharePoint, Teams and PowerBI.
Améliorez la qualité grâce à une plus grande collaboration
- Assurez-vous que tous les événements sont signalés dans l'eQMS avec n'importe quel appareil, même hors ligne.
- Impliquer facilement les parties prenantes internes/externes et de permettre des synergies interfonctionnelles.
Rentabilité et évolutivité
- Effet de levier total acquis Outils Microsoft 365 et bénéficiez d'un eQMS très rentable qui évolue avec votre entreprise.
- Toutes les parties prenantes internes et externes peuvent facilement alimenter le système de gestion de la qualité électronique sans frais supplémentaires.
Minimize project and IT risks with full ownership and control of your data
- Leverage your existing Microsoft Cloud and security as our eQMS is installed into your secure cloud environment – GCC High cloud-ready
- Bénéficier de technologies Microsoft fiables et de pointe qui évoluent en permanence.
Commencez tout de suite votre parcours de qualité efficace
- Facile et rapide à installer et à configurer dans votre environnement cloud Microsoft 365 sécurisé.
- Prebuilt modules and automation sont disponibles pour une mise en œuvre rapide et efficace du projet.
Libérer tout le potentiel de Microsoft 365 et de GenAI
- Efficiently interact with the eQMS while chatting on Teams, or with preconfigured Power Apps, robust automation and business intelligence (BI).
- Simplifiez les tâches quotidiennes et augmentez votre productivité grâce aux fonctions d'IA générative.
Atteindre l'excellence grâce à un système de gestion de la qualité de bout en bout
- Un abonnement qui intègre plus de 30 modules pour l'amélioration continue et la gestion du cycle de vie du produit.
- An intuitive and robust platform that facilitates compliance with any GxP, ISO, FDA, MDR, 21 CFR Part 11 requirement and more!
Faciliter la conformité grâce à l'automatisation et à la GenAI
- Faciliter la mise en conformité avec des schémas de processus visuels, des règlements et des processus clairement expliqués, et des signatures électroniques solides.
- Réduire le travail manuel et garantir l'exactitude avec des fonctions pré-intégrées d'automatisation des processus et d'IA générative.
Accelerate time to validation and stay compliant
- Deploy faster, stay compliant and seamlessly respond to audits with our Validation Toolkit providing a complete set of pre-defined, regulator-ready validation deliverables.
- Reduce the total cost of validation with a complete and audit-ready documentation suite curated by our expert validation partners.
Garantir des approbations valides grâce à des signatures électroniques solides
- Make sure only authorized people can sign documents/forms by using the trusted Microsoft multi-factor authentication system.
- Un moyen fiable d'approuver des procédures ou des dossiers techniques importants.
Satisfaire aux directives GxP et 21 CFR Part 11
- Document retention, access control et audit trail are included in BPAMedical365® to align with GxP and 21 CFR Part 11 requirements.
- Le module intégré de signature électronique offre une sécurité supplémentaire lors de l'approbation des documents.
Impliquer tous les utilisateurs dans le système de gestion de la qualité
- Faire de tous les utilisateurs des contributeurs actifs au SMQ avec leurs outils et dispositifs préférés (cols bleus et blancs, personnes sur le terrain, etc.).
- Ne modifiez pas les habitudes des utilisateurs et assurer l'adoption with SharePoint, Teams and PowerBI.
Améliorez la qualité grâce à une plus grande collaboration
- Assurez-vous que tous les événements sont signalés dans l'eQMS avec n'importe quel appareil, même hors ligne.
- Impliquer facilement les parties prenantes internes/externes et de permettre des synergies interfonctionnelles.
Rentabilité et évolutivité
- Effet de levier total acquis Outils Microsoft 365, as SharePoint, Teams and PowerBI, and benefit from a highly cost-effective eQMS that evolves with your business.
- Toutes les parties prenantes internes et externes peuvent facilement alimenter le système de gestion de la qualité électronique sans frais supplémentaires.
Minimize project and IT risks with full ownership of your data
- Leverage your existing Microsoft Cloud and security as our eQMS is installed into your secure cloud environment – GCC high cloud-ready
- Bénéficier de technologies Microsoft fiables et de pointe qui évoluent en permanence.
Commencez tout de suite votre parcours de qualité efficace
- Facile et rapide à installer et à configurer dans votre environnement cloud Microsoft 365 sécurisé.
- Prebuilt modules and automation sont disponibles pour une mise en œuvre rapide et efficace du projet.
Libérer tout le potentiel de Microsoft 365 et de GenAI
- Efficiently interact with the eQMS while chatting on Teams, or with preconfigured Power Apps, robust automation and business intelligence (BI).
- Simplifiez les tâches quotidiennes et augmentez votre productivité grâce aux fonctions d'IA générative.
Passer à une qualité intelligente avec BPAMedical365®
Le logiciel QMS de #1 est entièrement optimisé par Microsoft 365.

Des modules eQMS agiles pour tous vos besoins
Gestion des documents
Gestion du changement
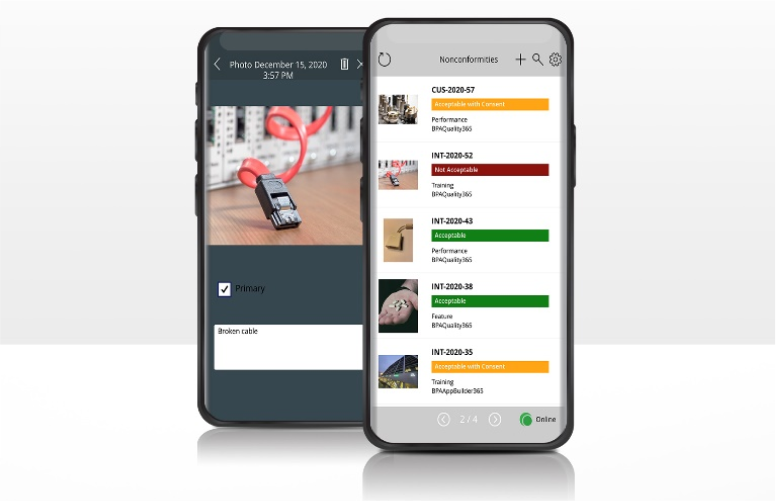
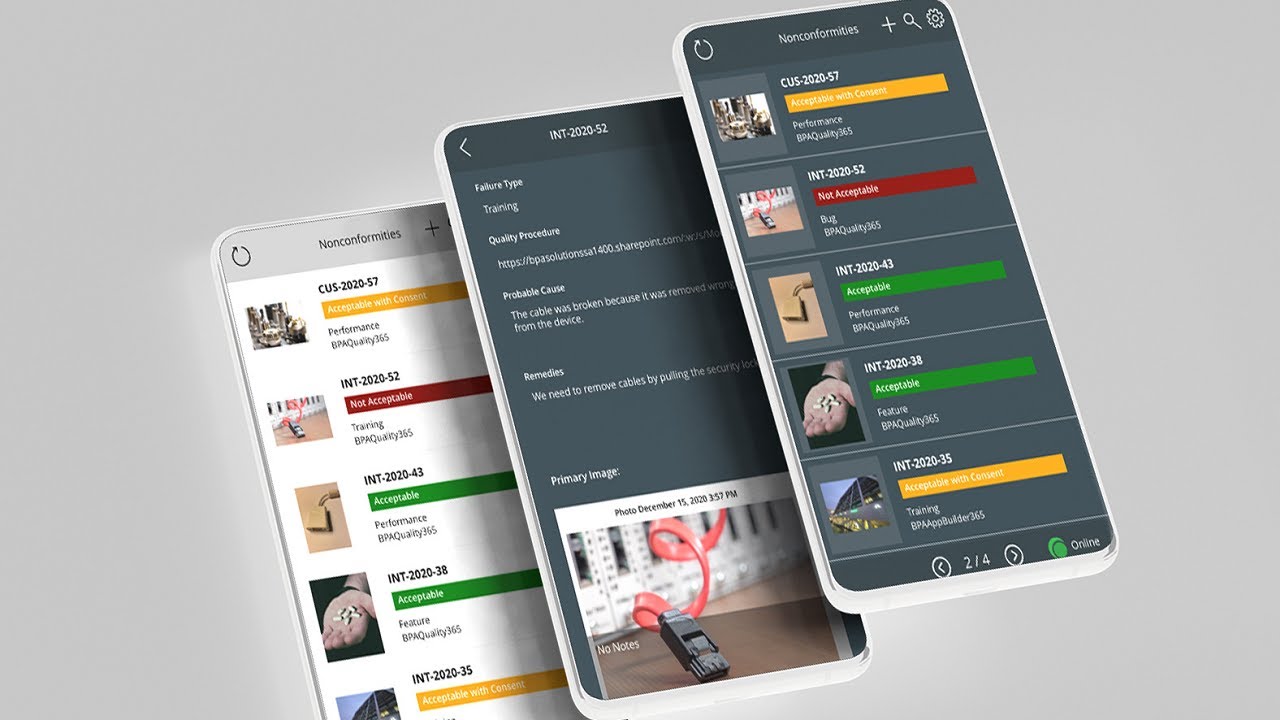
Gestion des non-conformités
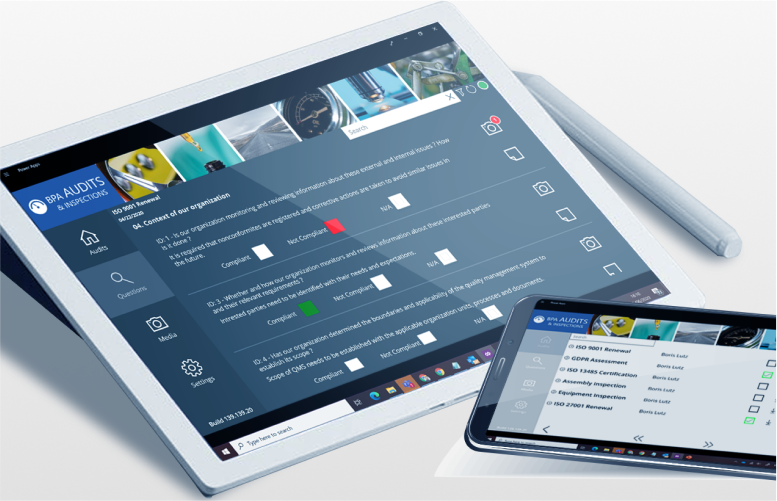
Gestion des audits
Gestion des processus
Gestion de la formation
Actions (CAPA)
Gestion des équipements
Gestion des risques
Gestion des fournisseurs
Santé et sécurité
Sécurité des informations
Aspects environnementaux
Request a demo
Voir d'autres vidéos de démonstration
Remplissez le formulaire et obtenez un accès exclusif à une variété de ressources vidéo sur nos solutions, modules, extensions, actualités et animations.
Check how BPAMedical365® travaux
Découvrez les principaux modules et fonctionnalités de l'eQMS de BPA

Visionnez des vidéos de démonstration supplémentaires pour les modules suivants :
- Gestion de documents
- Gestion des audits
- Gestion des non-conformités
Débloquer des performances de pointe avec Power Extensions
Vous souhaitez en savoir plus sur BPAMedical365®?
Frequently asked questions
Is BPAMedical365 compliant with regulatory requirements for Life Sciences and MedTech?
- Yes. BPAMedical365 is designed specifically for regulated industries and supports compliance with FDA, ISO, MDR, IVDR, and GxP expectations. The solution includes preconfigured workflows, structured documentation controls, secure eSignatures, and audit-ready records built directly on Microsoft 365.
How secure and legally compliant are BPA’s eSignatures for regulated environments?
- BPAMedical365 includes a secure, Microsoft-based eSignature module fully aligned with FDA 21 CFR Part 11 et EU Annex 11 requirements. Signatures are encrypted, traceable, tamper-evident, and validated with multi-factor authentication, making them ideal for validation, approvals, batch records, and controlled documentation.
Does BPAMedical365 support Product Lifecycle Management (PLM)?
- Yes. BPAMedical365 provides an integrated PLM module that lets you manage the entire product lifecycle (from early design and risk evaluation to manufacturing, post-market surveillance, and change control) ensuring full traceability across all documentation and quality processes.
Is the system validation-ready for FDA 21 CFR Part 11, EU Annex 11 and ISPE GAMP 5?
- Absolutely. BPAMedical365 includes a Validation Toolkit that provides SOP templates, test scripts, configuration documentation, and guidance aligned with industry-standard validation practices. This dramatically reduces validation time and effort for QA and Compliance teams.
Can BPAMedical365 be deployed in GCC High for highly regulated or government-related organizations?
- Yes. BPAMedical365 is fully compatible with Microsoft GCC High Cloud, offering a secure-by-design architecture that meets stringent data protection, sovereignty, and compliance requirements, ideal for U.S. public sector, defense, and contractors.
Where is my data stored, and who controls it?
- Since all your data remains inside your own Microsoft 365 tenant, BPA does not host or access your content. Microsoft provides the security, storage, and availability in accordance with major global regulations. You maintain full control over permissions and data access at all times, even during unexpected disruptions or after contract termination.
What does implementation look like? How long does it take? Which languages are supported?
- BPAMedical365 follows a standardized, efficient deployment approach for quick time-to-value. Most projects take 3–4 months with less than 10 days of BPA support, including weekly recorded sessions and knowledge transfer to make your team quickly autonomous.
- Prebuilt workflows, out-of-the-box modules, and online tutorials ensure a rapid, smooth go-live.
- Our eQMS is natively available in English, German, et French, ensuring seamless adoption across international teams. Additional languages can be added as part of an extended configuration, making the platform fully adaptable to global quality, compliance, and regulatory environments.
Can I start small and expand my BPAMedical365 environment later?
- Yes. The platform is highly scalable. Organizations can choose initial modules and expand at any time. Additional out-of-the-box modules, licenses, BI dashboards, and Power Extensions can be activated in minutes with no service disruption.
Does BPAMedical365 integrate with Microsoft Dynamics 365 and other ERP/CRM systems?
-
Absolutely. BPAMedical365 integrates natively with Microsoft Dynamics 365, including Business Central and Finance & Supply Chain Management (F&SCM), through our dedicated QMS connector. This enables synchronized records, automated workflows, unified master data, and real-time visibility across quality, operations, and ERP processes, creating a fully connected and efficient Microsoft-based ecosystem.
-
BPAMedical365 also integrates with third-party systems like SAP and major CRMs/ERPs. Because BPA runs on SharePoint and Microsoft 365, external integrations work like any standard SharePoint connection, supported by numerous connectors and customization options. Whether you need synced data, automated quality workflows, or deeper supply-chain connectivity, BPA helps you build a seamless, high-performance QMS environment tailored to your business.
What Microsoft 365 requirements do I need to run BPAMedical365?
- A standard Microsoft 365 subscription is required. Because BPAMedical365 runs natively within your Microsoft 365 environment, it leverages your existing security, Azure AD authentication, governance, and compliance tools, simplifying IT oversight while minimizing infrastructure complexity.
Accéder à des ressources supplémentaires pour BPAMedical365®
Remplissez le formulaire et bénéficiez d'un accès exclusif à notre site BPAMedical365® brochure de présentation.
Le logiciel QMS idéal pour votre marché spécifique
Agile - Evolutif - Sécurisé
Une solution éprouvée par l'industrie pour tous
- Gestionnaires de la qualité
- Département informatique
- Équipe de production
- Cadres de haut niveau
- Santé et sécurité
- Consultants
- Clients
- Fournisseurs
Simplifier la gestion de la qualité - Dites adieu aux systèmes papier inefficaces. Veillez à ce que les réglementations et les processus soient facilement compris et éliminez la confusion grâce à un logiciel de gestion de la qualité intuitif et intégré qui ne perturbe pas les habitudes.
Impliquer les utilisateurs dans l'amélioration continue - Avec une interface conviviale sur plusieurs appareils et plateformes (téléphones, ordinateurs, discussions en direct), BPAMedical365® encourage la collaboration en favorisant une culture d'amélioration continue à tous les niveaux hiérarchiques.
Renforcer la collaboration entre les entreprises - Faciliter la communication grâce à la messagerie instantanée, à la vidéoconférence et aux plates-formes de collaboration entre les équipes internes et externes. Renforcer le travail d'équipe grâce à une coordination efficace et au partage des connaissances.
Maintenir les certifications et réduire les coûts - Favorisez une culture de la précision et permettez à votre équipe qualité de se concentrer sur les questions stratégiques afin de préserver les certifications. BPAMédical365® tire parti de l'automatisation des processus de pointe pour accélérer les approbations, résoudre rapidement les problèmes et minimiser les coûts de non-conformité.
Faciliter la mise en conformité avec des réglementations complexes - Respecter la conformité en toute simplicité, quel que soit le secteur d'activité ou la région. BPAMédical365® est conçu pour répondre aux principes universels d'amélioration continue tout en adhérant à plusieurs réglementations en matière de qualité, de santé et de sécurité, de développement durable et de produits.
Augmenter la productivité - Gagnez du temps et des ressources avec BPAMedical365®Les fonctionnalités de GenAI : identifier rapidement les éléments ou documents similaires pour faciliter la résolution des problèmes et accélérer les processus avec Q-Pilot.
Adoptez la qualité/l’industrie 4.0 - L'avenir de la qualité est là, avec un logiciel QMS qui tire parti d'une automatisation robuste des processus, de fonctionnalités alimentées par l'IA et d'une intelligence économique avancée. Prospérez dans l'ère numérique et battez votre concurrence, en livrant des produits de qualité supérieure et en garantissant une conformité sans faille.
Assurer la continuité des activités - Exploitez toute la puissance de l'infrastructure en nuage de Microsoft 365 avec BPAMedical365®Le système de gestion de l'information de l'Union européenne (UE), avec une solide reprise après sinistre et des capacités optimales de continuité des activités (sauvegarde automatique des données, accessibilité en cas d'interruption).
Réduire les risques informatiques - BPAMedical365® offre une plateforme à 360° pour la gestion de la qualité, la billetterie, l'automatisation des processus, la formation et bien plus encore. Intégrée à Dynamics 365 et Azure Services, elle s'aligne sur votre stratégie de consolidation logicielle, minimisant ainsi les risques informatiques.
Garantir la sécurité et la confiance - Construit sur les fondations fiables de Microsoft 365 et hébergé dans votre cloud sécurisé Microsoft 365, BPAMedical365® s'aligne sur vos normes de sécurité, maximisant la disponibilité du système et la protection contre les cyber-attaques. Vos informations commerciales critiques ne pourraient pas se trouver dans un endroit plus sûr.
Maximiser le retour sur investissement - Éliminer les solutions redondantes, simplifier la gestion informatique et renforcer la sécurité avec BPAMedical365®. Cela se traduit par un retour sur investissement et des avantages immédiats, réduisant les coûts de déploiement par rapport aux solutions logicielles traditionnelles.
Exploiter les technologies existantes - Intégration transparente avec Microsoft 365, BPAMedical365® garantit une expérience utilisateur familière à travers les outils et les appareils. En tirant parti des répertoires d'utilisateurs et des mesures de sécurité existants, la mise en œuvre se fait sans heurts.
Rationaliser la maintenance des logiciels - BPA Solutions met automatiquement à jour ses applications et ses services, ce qui vous permet de toujours avoir accès aux dernières fonctionnalités, aux correctifs de sécurité et aux corrections de bogues sans intervention manuelle.
Améliorer la collaboration et la communication - Intégré à Microsoft Teams, BPAMedical365® alimente la collaboration en temps réel entre les équipes. Une solution qui rationalise la communication et la résolution des problèmes permet d'obtenir des résultats plus efficaces.
Détecter les problèmes à un stade précoce et minimiser les retouches - L'identification et la rectification des défauts à un stade précoce du processus de production sont essentielles au maintien des normes de qualité. Avec BPAMedical365® signaler à un stade précoce du processus toute anomalie et partager facilement les vidéos/procédures de bonnes pratiques afin de développer les compétences des collaborateurs
Normaliser et contrôler les processus de production - Appliquer facilement des processus standardisés avec BPAMedical365®Le contrôle des processus de production et l'identification des anomalies n'ont jamais été aussi faciles qu'aujourd'hui. Grâce aux applications Audit Power Apps de BPA, il n'a jamais été aussi facile de contrôler les processus de production et d'identifier les anomalies.
Collaborer avec les fournisseurs - Améliorez la cohérence et réduisez les perturbations en intégrant mieux les fournisseurs dans vos processus. Avec BPAMedical365® envoyez instantanément des messages à vos fournisseurs ou organisez des vidéoconférences avec eux, et intégrez-les facilement dans des flux de travail automatisés.
Développer les compétences des collaborateurs - Pour garantir des conditions de concurrence équitables entre les travailleurs, BPAMedical365® inclut des modules de formation pour renforcer les compétences des collaborateurs et automatiser entièrement les processus de formation aux documents.
Faciliter la gestion des équipements - BPAMedical365® rationalise la maintenance des équipements en s'appuyant sur les appareils mobiles et en intégrant un lecteur de codes-barres pour identifier facilement les équipements et signaler les anomalies.
Veiller au respect des réglementations - Dans les secteurs hautement réglementés, il est essentiel de garantir la conformité. BPAMédical365® fournit des outils permettant de vérifier facilement la conformité aux réglementations et d'éviter les amendes élevées, les problèmes juridiques et les atteintes à la réputation.
Adopter l'industrie 4.0 - L'avenir de la qualité est là, avec un eQMS qui tire parti d'une automatisation robuste des processus, de fonctionnalités alimentées par l'IA et d'une intelligence économique avancée. Prospérez dans l'ère numérique et battez votre concurrence, en livrant des produits de qualité supérieure et en garantissant une conformité sans faille.
Prendre en compte le retour d'information des clients et assurer leur satisfaction - Comprendre les besoins des clients et traiter les réclamations est essentiel pour la fidélité à la marque. BPAMedical365® simplifie la saisie des commentaires des clients et la réponse à ceux-ci, tout en permettant une détection rapide des anomalies, ce qui favorise la satisfaction des clients et la qualité.
Respecter la réglementation - BPAMedical365® facilite le respect des normes de qualité, des réglementations industrielles et des exigences légales dans tous les secteurs d'activité. En appliquant les principes universels d'amélioration continue, le logiciel répond à toutes les normes et réglementations.
Appliquer les contrôles internes - Le suivi de la réalisation des objectifs est essentiel pour la direction générale. BPAMedical365® comprend tous les modules nécessaires pour superviser et mettre en œuvre des contrôles internes efficaces.
Développer les compétences des employés -Pour garantir des conditions de concurrence équitables entre les travailleurs, BPAMedical365® comprend plusieurs modules permettant de renforcer les compétences et d'automatiser entièrement les processus de formation documentaire. Assurez-vous que les employés comprennent les procédures clés et testez facilement leurs connaissances avec BPAMedical365.®.
Maximiser le retour sur investissement - Éliminer les solutions redondantes, simplifier la gestion informatique et renforcer la sécurité avec BPAMedical365®. Obtenir un retour sur investissement et des avantages immédiats, en réduisant les coûts de déploiement par rapport aux solutions logicielles traditionnelles.
Rationaliser les processus et réduire les coûts - En automatisant les flux de travail, BPAMedical365® permet de réduire les coûts opérationnels et d'augmenter la rentabilité globale de l'organisation. Interagir avec le logiciel QMS en discutant en équipe accélère les processus et la prise de décision tout en réduisant le nombre de courriels encombrants.
Prendre des décisions rapides et éclairées - Vous avez du mal à consolider des données provenant de différentes sources ? Obtenez des informations exploitables et prenez des décisions éclairées avec BPAMedical365®L'entreprise fournit des tableaux de bord Power BI avancés permettant aux cadres de niveau C de contrôler efficacement les performances.
Respecter les réglementations en matière d'environnement, d'hygiène et de sécurité - BPAMedical365® comprend un module spécifiquement dédié à la santé et à la sécurité pour vous assurer que vous êtes en conformité avec les réglementations locales, nationales et internationales en matière de santé et de sécurité et pour faciliter la gestion des dossiers médicaux des employés et des évaluations médicales.
Signaler tous les incidents - Assurer la conformité avec les exigences réglementaires, les enquêtes en temps opportun et les actions correctives appropriées avec l'incident Power Apps de BPAMedical365.®. L'enregistrement des incidents et la déclaration des accidents du travail/maladies professionnelles aux autorités compétentes n'ont jamais été aussi rapides.
Identifier les dangers et évaluer les risques - Créer et tenir à jour des plans d'intervention d'urgence, faciliter l'identification et l'évaluation des risques et veiller à ce que les employés soient correctement préparés à réagir efficacement en cas d'urgence avec BPAMedical365®.
Évaluer les efforts en matière de sécurité - BPAMedical365® fournit des outils d'analyse de données et de reporting Power BI pour identifier les schémas d'accidents, ce qui permet aux départements EHS de mettre en œuvre des mesures proactives et de prévenir des incidents similaires.
Développer la formation et la sensibilisation - Les modules de formation et de compétences de BPAMedical365® est conçu pour garantir que l'ensemble du personnel est correctement formé pour exécuter ses tâches en toute sécurité et dans le respect des protocoles. Les utilisateurs peuvent interagir avec les modules EHS en équipe, ce qui accélère la prise de décision et renforce la coopération entre les services.
Surveiller les processus EHS - BPAMedical365® est l'outil ultime pour vérifier la performance des processus EHS. En rationalisant les inspections de sécurité grâce à Audit Power Apps, il permet aux professionnels de l'ESS de mener des évaluations approfondies et de suivre les actions correctives afin d'améliorer la sécurité sur le lieu de travail.
Responsabiliser les clients - Avec BPAMedical365®Les consultants d'EMCS peuvent travailler avec des organisations de différents secteurs, car ils prennent en charge diverses normes et réglementations. En accédant à des informations en temps réel, ils permettent à leurs clients de réaliser des améliorations efficaces et de se mettre en conformité de manière indépendante.
Collaboration sécurisée et transparente avec les clients - Les consultants externes peuvent collaborer de manière transparente avec les équipes internes de l'organisation en utilisant BPAMedical365.®. Grâce à leur accès sécurisé à la plateforme, ils peuvent facilement discuter dans Teams, collaborer sur des documents, assigner des tâches et partager rapidement des informations afin de prendre des décisions plus rapides sans avoir besoin de licence logicielle ou de courrier électronique.
Réduire les coûts et travailler efficacement - Les consultants peuvent accéder à BPAMedical365® avec un accès invité et des permissions sélectionnées. Grâce à son système robuste de gestion des tâches, les consultants peuvent travailler efficacement avec le client sur leurs activités en cours et accéder aux outils/données nécessaires, sans avoir besoin d'un logiciel supplémentaire.
Garantir la sécurité et la disponibilité des données - Hébergé dans le nuage Microsoft 365 de votre client, BPAMedical365® s'aligne sur les normes de sécurité de l'entreprise, garantissant la disponibilité du système et la protection contre 99% les cybermenaces. Les consultants et leurs clients bénéficient ainsi d'une sécurité optimale.
Utiliser des outils familiers pour rationaliser les processus - Exploiter les technologies Microsoft 365, BPAMedical365® est la plateforme la plus familière pour les consultants et leurs clients, facilitant l'adoption et simplifiant les processus d'audit/inspection : évaluer facilement la conformité, ajouter des constatations, des actions de suivi et rédiger automatiquement un rapport.
Développer des avantages concurrentiels uniques pour une croissance ultime - Avec BPAMedical365®Grâce à ce logiciel, les consultants élargissent leur offre de services et peuvent prendre en charge un plus grand nombre de clients dans différents secteurs d'activité. Le logiciel est très flexible pour refléter les méthodologies et les approches spécifiques des consultants : l'outil idéal pour développer des avantages concurrentiels uniques.
Obtenir un retour d'information de la part des clients - BPAMedical365® apporte des outils simples pour recueillir des commentaires et partager des enquêtes de satisfaction avec les clients, ce qui permet d'améliorer les relations client-fournisseur et la satisfaction des clients, sans licence logicielle supplémentaire.
Des outils familiers pour une collaboration sécurisée entre les organisations - Intégration transparente avec Microsoft 365, BPAMedical365® garantit une expérience utilisateur familière qui facilite l'adoption et préserve les habitudes. Les clients peuvent être invités en toute sécurité avec un accès invité et accéder facilement à des documents spécifiques avec des autorisations dédiées pour améliorer la collaboration entre les organisations et le partage des données (par exemple, partager des vidéos de bonnes pratiques, des documents de formation, etc.)
Rationaliser les flux de travail des clients - Pour rationaliser la collaboration avec les clients, des flux de travail automatisés inter-organisationnels peuvent être facilement configurés (par exemple, plaintes des clients, spécifications des clients, enquêtes de satisfaction, etc.) Vos équipes reçoivent des informations complètes et formatées pour une investigation et un traitement optimaux.
Traiter efficacement les réclamations des clients - BPAMedical365® facilite l'enregistrement des réclamations et permet de répondre efficacement au retour d'information des clients, ce qui favorise leur satisfaction. De solides flux de travail automatisés garantissent une réactivité rapide, ainsi qu'une notification et un suivi efficaces des actions correctives.
Économiser des coûts - Grâce à l'accès réservé aux invités et aux autorisations définies, les clients peuvent accéder aux outils et aux données nécessaires sans avoir à investir dans leur propre logiciel.
Qualification efficace des fournisseurs - BPAMedical365® rationalise radicalement le processus de qualification des fournisseurs, en facilitant leur enregistrement, en automatisant le processus d'approbation, en fournissant les informations nécessaires et en notifiant automatiquement les personnes concernées.
Rationaliser les audits des fournisseurs - BPAMedical365® simplifie le processus d'audit des fournisseurs, ce qui leur permet de se préparer et de participer plus facilement aux audits. Les questionnaires d'audit peuvent être générés automatiquement et partagés avec les fournisseurs, ce qui accélère le processus d'audit et d'inspection.
Automatiser le processus SCAR et consolider les flux de travail des fournisseurs - Lorsqu'une non-conformité est identifiée, des flux de travail automatisés inter-organisations attribuent des demandes d'actions correctives (SCAR) aux fournisseurs (par exemple, des demandes de déviation du fournisseur, des documents de spécification), rationalisant ainsi le processus de résolution des problèmes 8D et favorisant une relation plus forte et collaborative tout en minimisant les échanges de courrier inefficaces.
Assurer une collaboration sécurisée avec les fournisseurs - Avec BPAMedical365® Les fournisseurs peuvent accéder en toute sécurité aux documents et exigences de conformité via le logiciel, ce qui garantit clarté et transparence. Puisqu'elle exploite Microsoft 365 et Teams, la plateforme familière encourage l'adoption et la collaboration inter-organisationnelle. Hébergé dans votre cloud Microsoft 365 sécurisé, BPAMedical365® garantit la disponibilité du système et la protection contre 99% les cybermenaces. Vos fournisseurs bénéficient ainsi d'une sécurité optimale.
Réduire les coûts - Comme les fournisseurs bénéficient d'un accès invité gratuit à des formulaires et espaces de travail spécifiques avec des autorisations sélectionnées, ils peuvent accéder aux outils et à la documentation nécessaires sans avoir à investir dans un logiciel supplémentaire.
Microsoft 365
La meilleure technologie pour votre SMQ
Sécurisé
Microsoft 365 provides the highest security for your QMS data. You have full ownership and control of your data
De confiance
Rentable
Lower costs leveraging acquired Microsoft 365 tools for your QMS, as SharePoint, Teams, PowerBI, etc.
La confiance de plus de 200 clients satisfaits
qui ne cessent de croître avec BPA Solutions
Uppsala Monitoring Center
Uppsala Monitoring Center and BPA Solutions | #1 eQMS powered by Microsoft 365
Nos dernières nouvelles
Voyez par vous-même ce que BPA Solutions a à offrir
"Nous avons pu intégrer avec succès nos 1200 documents de qualité dans un système électronique entièrement géré, avec contrôle des versions et signatures électroniques, ce que nous devons mettre en œuvre pour répondre à nos exigences réglementaires....."