Calculate Your Cost Savings with a Digital Solution

It’s not easy to deliver engaging, efficient experiences when you’re held back by pen-to-paper-based processes.
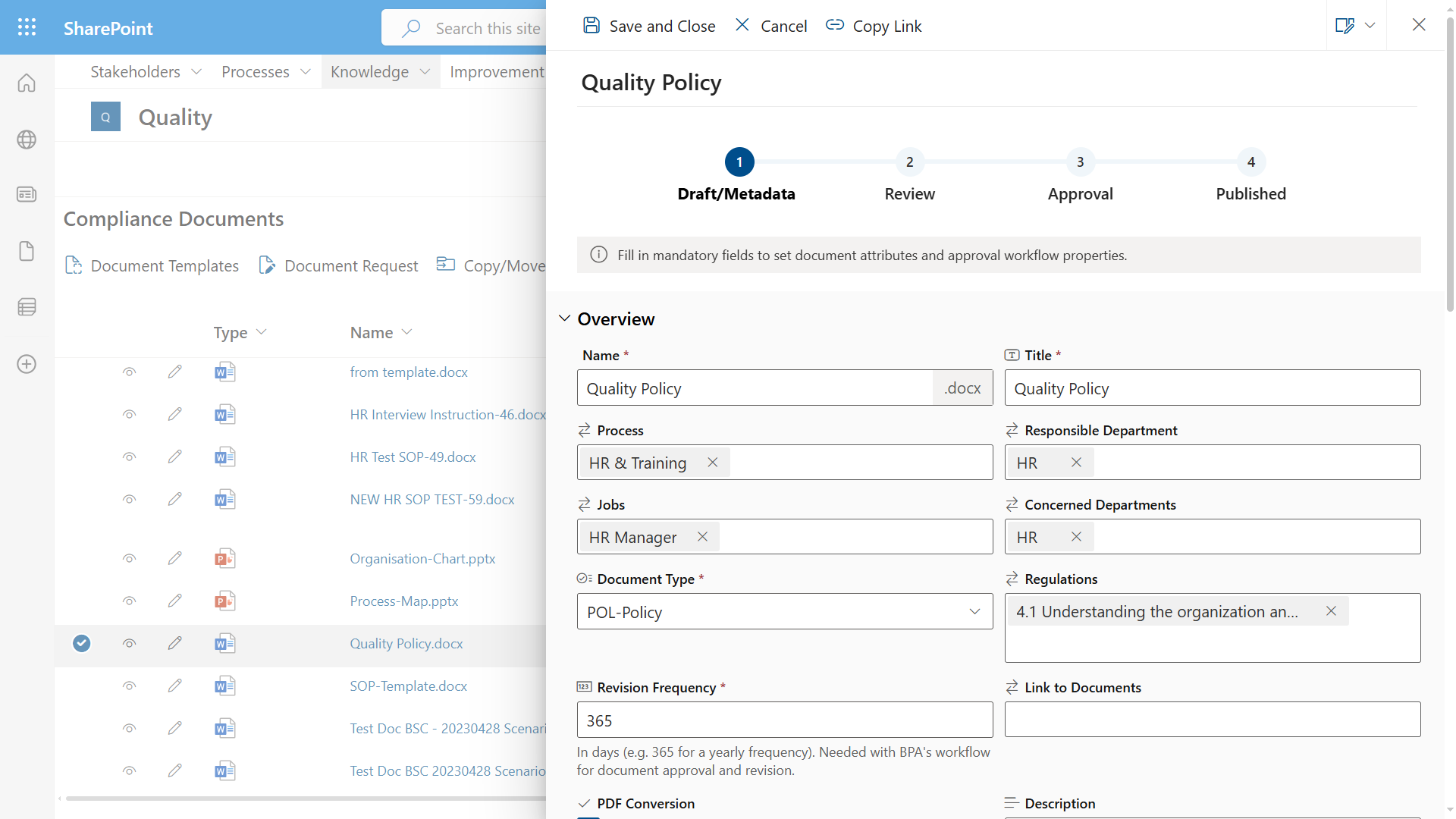
A digital solution offers a centralized location where organizations can efficiently store, organize, manage, access, and distribute information.

For the quality/regulatory department, information typically includes forms (audits, incidents, non-conformances, CAPA, indicators, risks, equipment maintenance, training, etc.) and quality/regulatory documents. By giving collaborators quick and easy access to the information they need anytime and anywhere, digital systems help employees focus more time on their core activities.
What are cost saving opportunities with a digital regulatory compliance solution?
- Electronic forms enforce collaborators to enter consistent information quickly and easily, reducing errors and manual rework.
- Automated workflows accelerate the process of creating and distributing forms and documents across devices and platforms.
- A digital system prevents collaborators to retype data at some point during the process thanks to data relations and automated reporting tools.
- No paper processing is needed anymore, saving printing, scanning, postage and archiving costs.
- Users spend a huge amount of time each day searching for information — an average of 37 minutes or 8% of the work day. Digital search tools can reduce search time by up to 50%.